GB/T 17593.1-2006 Textiles—Determination of heavy metals—Part 1: Atomic absorption spectrophotometry
Foreword
GB/T 17593 "Textiles—Determination of heavy metals" consists of the following parts:
— Part 1: Atomic absorption spectrophotometry;
— Part 2: Inductively coupled plasma atomic emission spectrometry;
— Part 3: Chromium (VI) by spectrophotometry;
— Part 4: Arsenic and mercury by atomic fluorescence spectrophotometry.
This part is Part 1 of GB/T 17593.
This part is a revision of GB/T 17593-1998 "Textiles—Test method of heavy metal ions—Atomic absorption spectrophotometry". Compared with GB/T 17593-1998, the main changes in this part are as follows:
— Added the determination of antimony (Sb) by graphite furnace atomic absorption spectrometry;
— Added the determination of copper (Cu) and antimony (Sb) by flame atomic absorption spectrometry;
— Changed the term "free amount of heavy metals" in the original standard to "extractable heavy metals";
— Deleted the determination of total heavy metal content;
— Deleted the methods for sample extraction using alkaline perspiration and saliva;
— Simplified the sample extraction procedure;
— Re-standardized the preparation methods for standard stock solutions of each element to be determined;
— Added the limit of determination for each element to be determined;
— Added an informative appendix recommending the use of matrix modifiers for graphite furnace atomic absorption spectrometry.
This part replaces GB/T 17593-1998.
Appendix A of this part is informative.
This part was proposed by the China National Textile Industry Council.
This part is under the jurisdiction of the Subcommittee on Basic Standards of the National Technical Committee on Textiles Standardization (SAC/TC 209/SC 1).
Drafting organizations of this part: Tianjin Entry-Exit Inspection and Quarantine Bureau of the People's Republic of China, Southern Testing Center of Shanghai Textile Research Institute.
Main drafters of this part: Guo Wei, Yu Tao, Yan Juan, Tu Yansheng, Zhu Naitong, Chen Yun.
Textiles—Determination of heavy metals—Part 1: Atomic absorption spectrophotometry**
WARNING — Personnel using this part of GB/T 17593 should have practical experience in a formal laboratory setting. This part does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user to establish appropriate safety and health practices and ensure compliance with any national regulatory conditions.
1 Scope
This part of GB/T 17593 specifies the method for the determination of extractable heavy metals including cadmium (Cd), cobalt (Co), chromium (Cr), copper (Cu), nickel (Ni), lead (Pb), antimony (Sb), and zinc (Zn) in textiles using graphite furnace or flame atomic absorption spectrophotometry.
This part is applicable to textile materials and products.
2 Normative References
The following referenced documents are indispensable for the application of this document. For dated references, only the edition cited applies. For undated references, the latest edition of the referenced document (including any amendments) applies.
GB/T 3922 Textiles—Tests for colour fastness—Colour fastness to perspiration (GB/T 3922-1995, eqv ISO 105-E04:1994)
GB/T 6682 Water for analytical laboratory use—Specification and test methods (GB/T 6682-1992, neq ISO 3696:1987)
3 Principle
The test specimen is extracted with an acid perspiration solution. Using atomic absorption spectrophotometry at the corresponding wavelengths, the absorbance of Cd, Co, Cr, Cu, Ni, Pb, Sb in the extract is measured with a graphite furnace atomic absorption spectrophotometer, and the absorbance of Cu, Sb, Zn is measured with a flame atomic absorption spectrophotometer. The content of each corresponding heavy metal ion is determined by comparison against a standard calibration curve. The content of extractable heavy metals in textiles obtained with acid perspiration solution is calculated.
4 Reagents and Materials
Unless otherwise specified, only reagents of guaranteed reagent grade and Grade 2 water as specified in GB/T 6682 shall be used.
4.1 Acid perspiration solution
Prepare the acid perspiration solution according to the requirements of GB/T 3922. The solution should be freshly prepared.
4.2 Single-element standard stock solutions
Standard stock solutions for each element can be purchased as certified reference materials or prepared as follows.
4.2.1 Cadmium (Cd) standard stock solution (1000 μg/mL)
Weigh 0.203 g of cadmium chloride (CdCl₂·5/2H₂O), dissolve in water, transfer to a 1000 mL volumetric flask, and dilute to the mark.
4.2.2 Cobalt (Co) standard stock solution (1000 μg/mL)
Weigh 2.630 g of anhydrous cobalt sulfate [obtained by igniting cobalt sulfate heptahydrate (CoSO₄·7H₂O) at 500°C–550°C to constant weight]. Add 150 mL of water, heat to dissolve, cool, transfer to a 1000 mL volumetric flask, and dilute to the mark.
4.2.3 Chromium (Cr) standard stock solution (1000 μg/mL)
Weigh 0.283 g of potassium dichromate (K₂Cr₂O₇), dissolve in water, transfer to a 1000 mL volumetric flask, and dilute to the mark.
4.2.4 Copper (Cu) standard stock solution (1000 μg/mL)
Weigh 0.393 g of copper sulfate pentahydrate (CuSO₄·5H₂O), dissolve in water, transfer to a 1000 mL volumetric flask, and dilute to the mark.
4.2.5 Nickel (Ni) standard stock solution (1000 μg/mL)
Weigh 0.448 g of nickel sulfate hexahydrate (NiSO₄·6H₂O), dissolve in water, transfer to a 1000 mL volumetric flask, and dilute to the mark.
4.2.6 Lead (Pb) standard stock solution (1000 μg/mL)
Weigh 0.160 g of lead nitrate [Pb(NO₃)₂], dissolve in 10 mL of nitric acid solution (1+9), transfer to a 1000 mL volumetric flask, and dilute to the mark.
4.2.7 Antimony (Sb) standard stock solution (1000 μg/mL)
Weigh 0.274 g of potassium antimony tartrate hemihydrate (C₄H₄KO₇Sb·1/2H₂O), dissolve in hydrochloric acid solution (10%), transfer to a 1000 mL volumetric flask, and dilute to the mark with hydrochloric acid solution (10%).
4.2.8 Zinc (Zn) standard stock solution (1000 μg/mL)
Weigh 0.440 g of zinc sulfate heptahydrate (ZnSO₄·7H₂O), dissolve in water, transfer to a 1000 mL volumetric flask, and dilute to the mark.
Note: Unless otherwise specified, standard stock solutions are stable for six months when stored at room temperature (15°C–25°C). They should be re-prepared if turbidity, precipitation, or color change occurs.
4.3 Standard working solution (10 μg/mL)
As needed, pipette appropriate amounts of one or more of the Cd, Cr, Cu, Ni, Pb, Sb, Zn, Co standard stock solutions into a 100 mL volumetric flask containing 5 mL of concentrated nitric acid. Dilute to the mark with water and mix well to prepare a single-element or multi-element standard working solution with a concentration of 10 μg/mL.
Note: This solution is valid for one week. It should be re-prepared if turbidity, precipitation, or color change occurs.
5 Apparatus and Instruments
5.1 Graphite furnace atomic absorption spectrophotometer: equipped with hollow cathode lamps for Cd, Co, Cr, Cu, Ni, Pb, Sb.
5.2 Flame atomic absorption spectrophotometer: equipped with hollow cathode lamps for Cu, Sb, Zn.
5.3 Conical flasks with stoppers: 150 mL.
5.4 Constant temperature water bath shaker: (37 ± 2)°C, shaking frequency 60 cycles/min.
6 Analysis Procedure
6.1 Preparation of the extract solution
Take a representative sample, cut it into pieces smaller than 5 mm × 5 mm, and mix well. Weigh 4 g of the specimen in duplicate (for parallel testing), accurate to 0.01 g, into a conical flask with stopper (5.3). Add 80 mL of acid perspiration solution (4.1), ensuring the fibers are thoroughly wetted. Place the flask into the constant temperature water bath shaker (5.4) and shake for 60 minutes. Remove the flask, let it stand and cool to room temperature, then filter. The filtrate is the sample solution for analysis.
6.2 Determination
6.2.1 Dilute the standard working solution (4.3) stepwise with water to prepare a series of working solutions of appropriate concentrations. Using the graphite furnace atomic absorption spectrophotometer, measure the absorbance of Cd, Co, Cr, Cu, Ni, Pb, Sb in the series of working solutions in order of increasing concentration at wavelengths 228.8 nm (Cd), 240.7 nm (Co), 357.9 nm (Cr), 324.7 nm (Cu), 232.0 nm (Ni), 283.3 nm (Pb), and 217.6 nm (Sb), respectively. Alternatively, using the flame atomic absorption spectrophotometer, measure the absorbance of Cu, Sb, Zn in the series of working solutions in order of increasing concentration at wavelengths 324.7 nm (Cu), 217.6 nm (Sb), and 213.9 nm (Zn), respectively. Plot the calibration curves with absorbance on the y-axis and element concentration (μg/mL) on the x-axis.
6.2.2 Using the instrument conditions set in 6.2.1 and the corresponding wavelengths, measure the absorbance of each element in the blank solution and the sample solution (6.1). Determine the concentration of each element from the corresponding calibration curve.
Note: To achieve good limits of detection and precision, it is recommended to use matrix modifiers when determining Cd, Co, Cr, Cu, Ni, Pb, and Sb by graphite furnace atomic absorption spectrophotometry. Refer to Appendix A for graphite furnace parameters and matrix modifier suggestions.
7 Calculation of Results
The content of extractable heavy metal element *i* in the specimen is calculated according to equation (1):

( X_i = rac{(c_i - c_0) imes V imes F}{m} )
Where:
( X_i ) — Content of extractable heavy metal element *i*, in milligrams per kilogram (mg/kg);
( c_i ) — Mass concentration of element *i* in the sample solution, in micrograms per milliliter (μg/mL);
( c_0 ) — Mass concentration of element *i* in the blank solution, in micrograms per milliliter (μg/mL);
( V ) — Total volume of the extract solution, in milliliters (mL) (here V = 80 mL);
( F ) — Dilution factor, if any (if no dilution, F=1);
( m ) — Mass of the specimen, in grams (g).
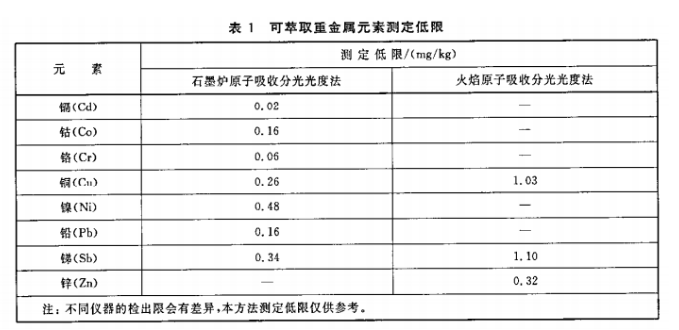
8 Limit of Determination and Precision
8.1 Limit of determination
The limits of determination for this method are given in Table 1.
(Table 1 content placeholder)
8.2 Precision
For two independent test results obtained in the same laboratory, by the same operator using the same equipment, following the same test method on the same test material within a short time interval, the absolute difference between the two results should not exceed 10% of the arithmetic mean of the two values. This statement is made with a 5% risk of the difference exceeding 10% of the arithmetic mean of the two results.
9 Test Report
The test report shall contain the following information:
a) Reference to this part;
b) Description of the sample;
c) Instrument used;
d) Date of the test;
e) Content of each heavy metal in the sample;
f) Any deviation from this part.
Appendix A
(Informative)
Parameters for Graphite Furnace (Transverse Heated Platform Tube) and Matrix Modifiers
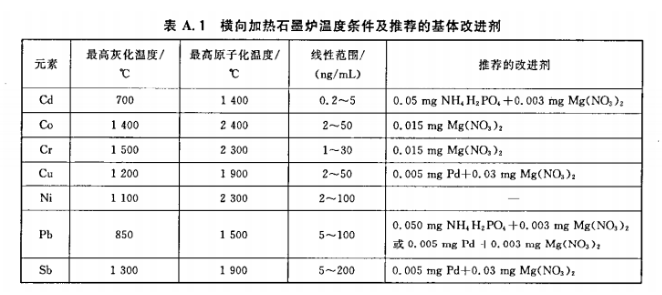
A.1 Graphite furnace parameters and matrix modifiers
Table A.1 provides reference temperature conditions for a transverse heated graphite furnace. Other instrument models may use these as a guide. A typical sample injection volume is 10 μL, followed by 5 μL of matrix modifier.
(Table A.1 content placeholder)
A.2 Preparation method for matrix modifier
Calculate the matrix modifier amount according to reference formula (A.1).
(Formula A.1 content placeholder)

Source: National Standard of the People's Republic of China GB/T 17593.1-2006 Replaces GB/T 17593-1998
