GB/T 30157-2013 Textiles—Determination of total lead and total cadmium content
Foreword
This standard was drafted in accordance with the rules given in GB/T 1.1-2009.
This standard was proposed by the China National Textile and Apparel Council.
This standard is under the jurisdiction of the Subcommittee on Basic Standards of the National Technical Committee on Textiles Standardization (SAC/TC 209/SC 1).
Drafting organizations of this standard: China Textile Standard (Beijing) Inspection and Certification Center Co., Ltd., National Center for Quality Supervision and Inspection of Textile Products, Ningbo Entry-Exit Inspection and Quarantine Bureau of the People's Republic of China.
Main drafters of this standard: Zhang Hui, Jing Tingting, Zheng Yuxiu, Bao Qibei, Feng Yun, Fu Kejie.
Determination of Total Lead and Total Cadmium Content in Textiles
Warning: Personnel using this standard should have practical experience in a formal laboratory setting. This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user to establish appropriate safety and health practices and ensure compliance with any national regulatory conditions.
1 Scope
This standard specifies the method for the determination of total lead and total cadmium content in textile products.
This standard is applicable to all types of textile products.
2 Normative References
The following documents are indispensable for the application of this document. For dated references, only the edition cited applies. For undated references, the latest edition (including any amendments) applies.
GB/T 6682 Water for analytical laboratory use — Specification and test methods
3 Principle
The test specimen is digested with concentrated acid. After digestion, the solution is diluted to a fixed volume. The emission intensity of lead and cadmium is measured under appropriate conditions using an Inductively Coupled Plasma Atomic Emission Spectrometer (ICP-AES), or the absorbance is measured using an Atomic Absorption Spectrophotometer (AAS). The concentration of each metal ion is determined by comparing against a standard calibration curve, and the total amount of heavy metals in the specimen is calculated.
4 Reagents and Materials
Unless otherwise specified, only reagents of analytical grade or higher, and Grade 2 water or higher as specified in GB/T 6682, shall be used.
4.1 Concentrated nitric acid (HNO₃)
4.2 3% (volume fraction) nitric acid: Pipette 3 mL of concentrated nitric acid (4.1) into a 100 mL volumetric flask and dilute to the mark with Grade 2 water.
4.3 Fluoroboric acid (HBF₄)
4.4 Hydrofluoric acid (HF)
4.5 Hydrogen peroxide (H₂O₂)
4.6 Standard stock solutions: Standard stock solutions for each element can be purchased as certified reference materials or prepared as follows.
4.6.1 Lead (Pb) standard stock solution (1000 μg/mL)
Weigh 0.160 g of lead nitrate [Pb(NO₃)₂], dissolve in 10 mL of nitric acid (1+9), transfer to a 100 mL volumetric flask, and dilute to the mark.
4.6.2 Cadmium (Cd) standard stock solution (1000 μg/mL)
Weigh 0.203 g of cadmium chloride hydrate (CdCl₂·H₂O), dissolve in 10 mL of nitric acid (1+9), transfer to a 100 mL volumetric flask, and dilute to the mark.
Note: Unless otherwise specified, standard stock solutions are stable for 6 months when stored at room temperature (15°C–25°C). They should be re-prepared if turbidity, precipitation, or color change occurs.
4.7 Standard working solution (50 μg/mL)
Accurately pipette 2.50 mL of the standard stock solution into a volumetric flask and dilute to 50 mL with 3% nitric acid (4.2).
Note: The standard working solution is generally stable for two weeks at room temperature (15°C–25°C). It should be re-prepared if turbidity, precipitation, or color change occurs.
5 Apparatus and Instruments
5.1 Inductively Coupled Plasma Atomic Emission Spectrometer (ICP-AES) or Atomic Absorption Spectrophotometer (AAS)
5.2 Microwave digestion system: with programmable temperature control.
5.3 Digestion vessels
5.4 Disposable blades
5.5 Volumetric flasks: 50 mL, 100 mL
5.6 Pipettes: 0.5 mL, 1.0 mL, 2.0 mL
5.7 Aqueous phase filtration membrane: pore size 0.45 μm
5.8 Analytical balance: accuracy 0.01 mg
6 Analysis Procedure
6.1 Specimen preparation and processing
6.1.1 Preparation
Take a representative specimen and cut it into small pieces (approx. 5 mm × 5 mm). Weigh approximately 0.2 g of the specimen, accurate to 0.0001 g.
6.1.2 Digestion
Add 5.0 mL of concentrated nitric acid (4.1) to the digestion vessel containing the test specimen and to an empty vessel (blank). Allow the sample and acid to react completely at room temperature. Seal the vessels and place them in the microwave digestion system (5.2). Heat to (175 ± 5)°C over 10 minutes and hold at (175 ± 5)°C for 5 minutes. Allow the vessels to cool for at least 5 minutes inside the system, then remove them. Before opening, cool the vessels in a fume hood to room temperature or for at least 30 minutes.
Note 1: Different microwave systems may require different programs. The key is to ensure complete digestion of the sample.
Note 2: For difficult-to-digest coated samples (e.g., PU), appropriate amounts of fluoroboric acid, hydrofluoric acid, or hydrogen peroxide may be added.
Note 3: If hydrofluoric acid is used, add 30 mL of 4% boric acid to each vessel after digestion to complex the fluoride and protect the quartz torch of the ICP.
6.1.3 Solution making up
Transfer the digested solution quantitatively into a 50 mL volumetric flask (5.5). Rinse the digestion vessel three times with small amounts of water and combine the rinsings into the volumetric flask. Dilute to the mark with water and mix thoroughly. Filter the solution through an aqueous phase filtration membrane (5.7). The filtrate should be analyzed promptly by ICP-AES or AAS (5.1).
Note: See Appendix A for the pretreatment method for accessories and decorative parts of textile products.
6.2 ICP-AES Analysis and Determination
6.2.1 Analytical wavelengths for elements by ICP-AES
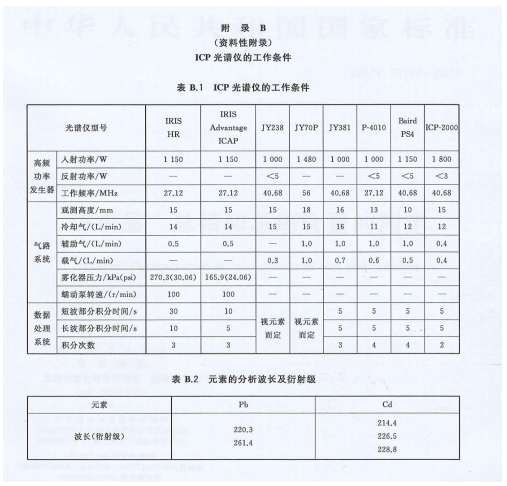
Pb: 220.3 nm; Cd: 214.4 nm.
Note: To eliminate spectral interference from other elements, an additional spectral line at another wavelength can be selected as a reference.
6.2.2 Determination
6.2.2.1 Calibration curve determination
According to test requirements and instrument conditions, dilute the standard working solution (4.7) with water stepwise to prepare a series of working solutions of appropriate concentrations. Set the instrument conditions. Ignite the plasma. Once stable, measure the spectral intensity of the elements in the series of working solutions in order of increasing concentration. Plot the calibration curve with spectral intensity on the y-axis and element concentration (μg/mL) on the x-axis.
6.2.2.2 Sample solution determination
Using the instrument conditions set in 6.2.2.1, measure the spectral intensity of the elements in the blank solution and the sample solution (6.1.3). Determine the concentration of each element from the calibration curve.
Note 1: If the concentration of lead or cadmium in the sample solution exceeds 1.5 times the highest point of the calibration curve, the solution should be appropriately diluted and re-analyzed.
Note 2: Flame Atomic Absorption Spectrometry (FAAS) can also be used for determining lead and cadmium concentrations. When using this method, attention should be paid to correcting possible interferences, and the method used must be specified in the report.
Note 3: Instrument conditions may vary. See Appendix B for working conditions of some ICP spectrometers and analytical wavelengths for elements.
6.2.2.3 Quality control
A quality control check should be performed after every 20 tests by measuring a calibration standard to ensure the accuracy of results.
7 Calculation of Results
7.1 The content of heavy metal element *i* in the specimen is calculated according to equation (1):
Wi = [(Ci - C0) × V] / m

Where:
Wi — Total content of heavy metal element *i* in the specimen, in milligrams per kilogram (mg/kg);
Ci — Mass concentration of heavy metal element *i* in the sample solution, in micrograms per milliliter (μg/mL);
C0 — Mass concentration of heavy metal element *i* in the blank solution, in micrograms per milliliter (μg/mL);
V — Total volume of the sample solution, in milliliters (mL);
m — Mass of the specimen, in grams (g).
7.2 The result is rounded to the nearest whole number. If the result is below the limit of determination, it is reported as "not detected".
8 Limit of Determination and Precision
8.1 Limit of detection of the method
The limit of detection for lead (Pb) by this method is 2.50 mg/kg. The limit of detection for cadmium (Cd) by this method is 0.25 mg/kg. If FAAS is used, the detection limits may differ.
8.2 Precision
For two independent test results obtained in the same laboratory, by the same operator using the same equipment, following the same test method on the same test material within a short time interval, the relative standard deviation is not greater than 10%. This statement is made with a 5% risk of the difference exceeding 10% of the arithmetic mean of the two results.
9 Test Report
The test report shall contain at least the following information:
a) Reference to this standard (i.e., the number of this standard);
b) Source and description of the sample;
c) Test results;
d) Any details of deviation from this standard;
e) Date of the test.
Appendix A
(Informative)
Pretreatment Method for Accessories and Decorative Parts Used in Textile Products
A.1 Reagents
A.1.1 Reagents listed in Clause 4.
A.1.2 Concentrated hydrochloric acid (HCl).
A.2 Apparatus and Equipment
A.2.1 Instruments and equipment listed in Clause 5.
A.2.2 Metal cutter.
A.2.3 Rotary mill.
A.2.4 Cryogenic mill.
A.3 Analysis Procedure
A.3.1 Sample preparation
A.3.1.1 Preparation of coated samples
Scrape off the surface coating using a disposable blade (A.2.4), avoiding the substrate material as much as possible. Weigh 20 mg – 100 mg of the scraped coating as the test specimen, accurate to 0.1 mg, into a microwave digestion vessel (5.3). Proceed with processing according to 6.1.2 – 6.1.3.
A.3.1.2 Preparation of uncoated samples
A.3.1.2.1 Metallic materials
Cut the sample into small pieces using a metal cutter (A.2.2) or grind it using a rotary mill (A.2.3). Weigh 30 mg – 100 mg of the prepared specimen, accurate to 0.1 mg, into a microwave digestion vessel (5.3). Add 4.5 mL concentrated nitric acid (4.1) and 1.5 mL concentrated hydrochloric acid (A.1.2) to the vessels containing the test specimen and the blank. Allow the reaction to complete. Seal the vessels and place them in the microwave digestion system (5.2). Heat to (175 ± 5)°C over 5.5 minutes and hold at (175 ± 5)°C for 4.5 minutes. Allow to cool for at least 5 minutes inside the system, then remove. Before opening, cool in a fume hood to room temperature or for at least 30 minutes. Depending on the sample weight taken, transfer the digested solution quantitatively into a suitable volumetric flask (5.5). Rinse the digestion vessel with small amounts of water, combine the rinsings into the volumetric flask, dilute to the mark with water, and mix. Filter through an aqueous phase filtration membrane (5.7). This solution is ready for analysis by ICP-AES or AAS (5.1) following 6.2.
Note: Generally, for sample weights of 20 mg – 49 mg, transfer to a 10 mL volumetric flask. For sample weights of 50 mg – 100 mg, transfer to a 25 mL volumetric flask.
A.3.1.2.2 Non-siliceous materials (e.g., plastics, polymers)
Grind the sample using a cryogenic mill (A.2.4) or cut it into pieces not larger than 1 mm × 1 mm × 1 mm. Weigh 30 mg – 100 mg of the prepared specimen, accurate to 0.1 mg, into a microwave digestion vessel. Add 8.0 mL concentrated nitric acid (4.1) to the vessels containing the test specimen and the blank. Allow the reaction to complete. Seal the vessels and place them in the microwave digestion system (5.2). Heat to (210 ± 5)°C over 20 minutes and hold at (210 ± 5)°C for 10 minutes. Allow to cool for at least 5 minutes inside the system, then remove. Before opening, cool in a fume hood to room temperature or for at least 30 minutes. Depending on the sample weight taken, transfer the digested solution quantitatively into a suitable volumetric flask (5.5). Rinse the digestion vessel with small amounts of water, combine the rinsings into the volumetric flask, dilute to the mark with water, and mix. Filter through an aqueous phase filtration membrane (5.7). This solution is ready for analysis by ICP-AES or other suitable instrument (5.1) following 6.2.
A.3.1.2.3 Siliceous materials (e.g., crystal, glass)
Grind the sample using a cryogenic mill (A.2.4). Weigh 30 mg – 100 mg of the ground specimen, accurate to 0.1 mg, into a microwave digestion vessel. Add 3 mL concentrated nitric acid (4.1) and 1 mL hydrofluoric acid (4.4) to the vessels containing the test specimen and the blank. Allow the reaction to complete. Seal the vessels and place them in the microwave digestion system (5.2). Heat to (175 ± 5)°C over 5.5 minutes and hold at (175 ± 5)°C for 9.5 minutes. Allow to cool for at least 5 minutes inside the system, then remove. Before opening, cool in a fume hood to room temperature or for at least 30 minutes. *Note: After digestion with HF, add 30 mL of 4% boric acid to complex the fluoride before proceeding.* Depending on the sample weight taken, transfer the digested solution quantitatively into a suitable volumetric flask (5.5). Rinse the digestion vessel with small amounts of water, combine the rinsings into the volumetric flask, dilute to the mark with water, and mix. Filter through an aqueous phase filtration membrane (5.7). This solution is ready for analysis by ICP-AES or other suitable instrument (5.1) following 6.2.
Source: National Standard of the People's Republic of China GB/T 30157-2013*
